
Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Common Chemical Compounds of Chromium ReferencesĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.It gives rubies and emeralds their color. Also used in plating for car parts, tools, knives, camouflage paint, stereos, video tapes and lasers. Uses of Chromium: Used to make stainless steel.Primary mining sources are Turkey, South Africa, Zimbabwe, Russia and Philippines. Annual world wide production around 20,000 tons. Sources of Chromium: Does not occur free in nature.70kg human: 14 mg Who / Where / When / How Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Target Organs: Eyes, skin, respiratory system.Routes of Exposure: Inhalation Ingestion Skin and/or eye contact.Notes: The PEL also applies to insoluble chromium salts.Vapor Pressure = C Regulatory / Health.Flammablity Class: Non-combustible solid (except as dust).Enthalpy of Vaporization: 348.8 kJ/mole.Enthalpy of Atomization: 397.5 kJ/mole 25☌.Description: Hard brittle gray transition metal.Conductivity Electrical: 0.0774 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 6.2E -6.Valence Electron Potential (-eV): 170 Physical Properties of Chromium.Incompatibilities: Strong oxidizers (such as hydrogen peroxide), alkalis.Electronegativity: 1.66 (Pauling) 1.56 (Allrod Rochow).Electrochemical Equivalent: 0.32333g/amp-hr.Valence Electrons: 3d 5 4s 1 Electron Dot Model. 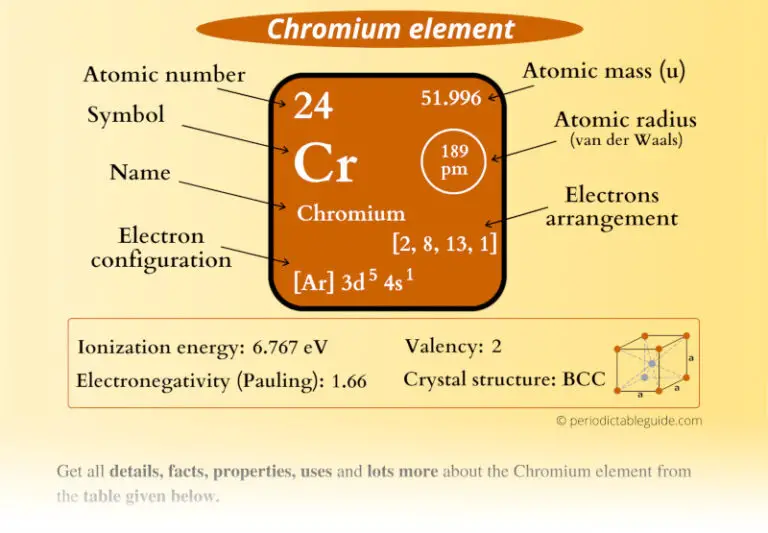
Number of Neutrons (most common/stable nuclide): 28.Number of Electrons (with no charge): 24.Electrons per Energy Level: 2,8,13,1 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 5 4s 1.Crystal Structure: Cubic body centered.Cross Section (Thermal Neutron Capture)σ a/ barns : 3.1.Swedish: Krom Atomic Structure of Chromium.Series: Transition Metals Chromium's Name in Other Languages.Common Chemical Compounds of Chromium Overview of Chromium.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Chromium - CrĬomprehensive data on the chemical element Chromium is provided on this page including scores of properties, element names in many languages, most known nuclides of Chromium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
